Exploring Hormone-Disrupting Chemicals and their impact with computational chemistry
Find out how our team at NovaMechanics, together with our experimental partners, used computational tools to understand how everyday chemicals interfere with our hormones – and what this means for your health.
What are Endocrine Disrupting Chemicals, and why should we care?
Every day, we are exposed to hundreds of synthetic chemicals that are present in the plastics of our food packaging, in personal care products, in the food we eat and can interfere with our body’s hormonal system. These compounds are called Endocrine Disrupting Chemicals, or EDCs.
Our hormones, molecules like estrogen and testosterone, act as messengers, instructing our cells how to grow, develop, and maintain metabolic balance. When an EDC mimics or blocks these hormones, it can set off a chain of disruptions that have been linked to metabolic diseases, including metabolic dysfunction-associated steatotic liver disease (fatty liver) and type 2 diabetes.
The NEMESIS project is dedicated to understanding exactly how this happens at the molecular level, so that scientists, regulators, and ultimately the public can make better-informed decisions about chemical safety.
Our focus: Bisphenols and Zearalenone
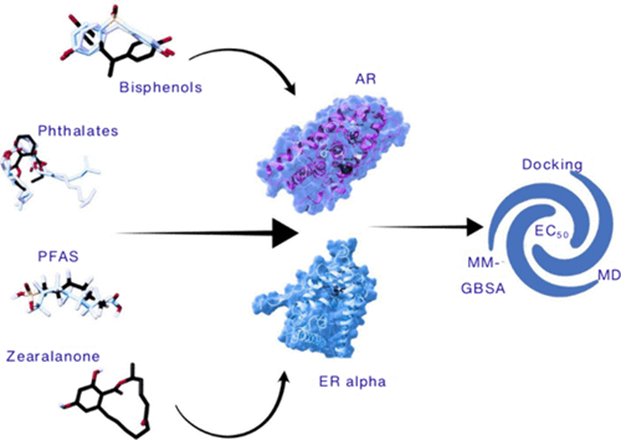
In a recently published study in the Journal of Chemical Information and Modeling, our team at NovaMechanics, together with partners from the Dr Margarete Fischer-Bosch Institute of Clinical Pharmacology in Stuttgart and the Karolinska Institutet in Stockholm, took a close look at two important groups of EDCs:
- Bisphenols: A family of industrial chemicals widely used in plastics (including the well-known Bisphenol A, or BPA), epoxy resins, and thermal paper receipts. Despite regulatory restrictions on BPA in many countries, its chemical cousins (BPS, BPF, and others) have proliferated as replacements, often with poorly understood health effects.
- Zearalenone: A mycotoxin produced naturally by certain fungi that contaminate grains such as wheat, corn, and barley. It is a known environmental estrogen that can enter the human food supply.
Both types of chemicals have been suspected of interfering with the estrogen receptor alpha (ERα) and the androgen receptor (AR), the cellular “antennas” that pick up estrogen and testosterone signals, respectively. But exactly how they bind to these receptors, and how strongly, was not fully understood. That’s what our study set out to answer.

Our approach: Connecting computers and laboratory experiments
One of the exciting things about modern chemical safety science is that we no longer have to test every chemical in a test tube or on animals. Using computational chemistry, we can simulate how molecules behave at the atomic scale on a computer and then validate the most important findings with targeted laboratory experiments.
Our study combined three powerful computational approaches with cell-based assays:
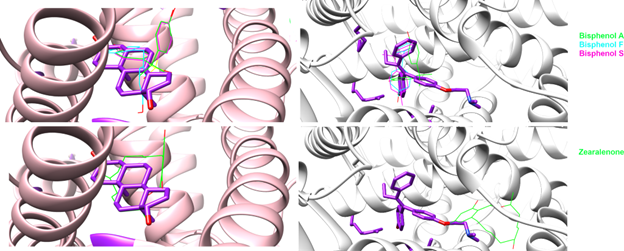
- Molecular Docking: Like a digital lock-and-key experiment, we used docking simulations to predict how each chemical “fits” into the binding pocket of ERα and AR. This gives us a snapshot of the initial interaction.
- Molecular Dynamics (MD) Simulations: Proteins are not rigid structures; they wiggle, flex, and breathe. We ran extensive molecular simulations of each chemical bound to its receptor to understand how the binding evolves over time and which interactions are most important.
- Binding Free Energy Calculations (MM-GBSA and Alchemical Methods): We estimated the actual strength of binding for each chemical — essentially asking “how tightly does this molecule bind to the receptor?”, using two computational methods.
- Cell-Based Inhibition Assays: Our experimental partners measured the actual biological effect of these chemicals in human cells (HEK293), confirming whether they activated or blocked the estrogen and androgen receptors.
All the computational workflows were built using the Asclepios Enalos KNIME Nodes; an open, user-friendly platform developed by NovaMechanics that makes advanced cheminformatics tools accessible to researchers without deep programming expertise using a no-code philosophy. This kind of open-science infrastructure is central to our mission: making the best science available to as many people as possible by alleviating any potential technical obstacles.
What Did We Find?
The results were illuminating and, in some cases, concerning. Both bisphenols and zearalenone showed increased binding to ERα, meaning they can potentially activate estrogen signaling pathways in cells, even at relatively low concentrations. This was confirmed by the laboratory experiments, where we saw dose-dependent activation of the receptor.
For the androgen receptor, the picture was one of antagonism: these chemicals bind to AR and block it, preventing the body’s natural androgens from doing their job. The MD simulations helped explain why, by revealing specific molecular interactions that stabilize the chemical inside the receptor’s binding pocket and interfere with the normal signalling mechanism.
Importantly, there were meaningful differences among individual bisphenol variants. Not all BPA replacements are alike. Some showed stronger binding than BPA itself, which is a finding that regulators and industry need to take seriously, as “regrettable substitution” (swapping one harmful chemical for another) remains a real problem.
Why is this important for your health?
The estrogen and androgen receptors are not just about reproduction. They play crucial roles in regulating metabolism, fat storage, liver function, and energy balance. When EDCs hijack or block these receptors, the downstream effects can contribute to exactly the kinds of metabolic diseases that NEMESIS is investigating: non-alcoholic fatty liver disease and type 2 diabetes.
Our study contributes important mechanistic evidence behind these health associations. By understanding exactly how bisphenols and zearalenone interact with nuclear hormone receptors, we help build the scientific foundation that regulators need to set better safety limits and develop more protective policies.
Open Science in Action
This work reflects the open-science principles at the heart of NEMESIS. The Asclepios Enalos KNIME workflows used in this study were developed to ensure the reproducible and transparent application of computational methods, utilizing open-source tools that the whole scientific community can benefit from. They can be used by anyone who is a non-expert in the field of cheminformatics or computational chemistry, without having prior programming expertise.
Moreover, we aim to publish in open-access journals so that the results of publicly funded research are available to everyone, from fellow scientists to policymakers to curious members of the public.
What’s next?
This study is one piece of a much larger puzzle. Within NEMESIS, we are continuing to develop and apply computational tools to map the full landscape of how metabolic-disrupting chemicals affect human health, across different receptors, different cell types, and different chemical mixtures. The ultimate goal is a comprehensive, predictive toolkit that can help identify dangerous chemicals before they reach consumers.
We will be sharing more results and insights as the project progresses. In the meantime, you can follow NEMESIS on LinkedIn and check back here for more blog posts from partners across the consortium.
Read the full paper
Videos
Chemical Imposters: The Hidden Science of Endocrine Disruptors (YouTube)
The molecular secrets of hormone disruptors (YouTube)
